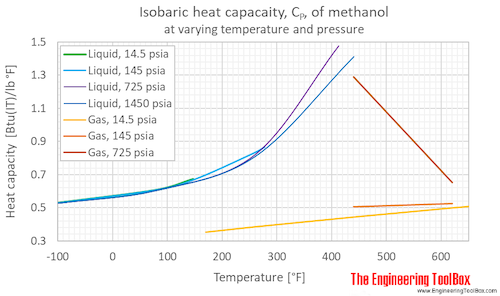
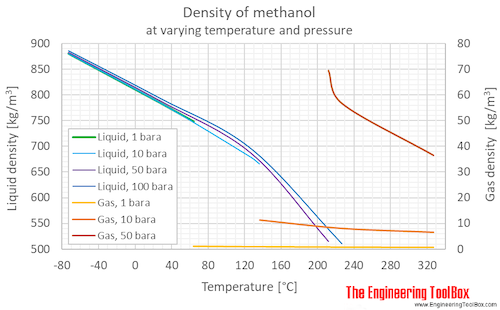
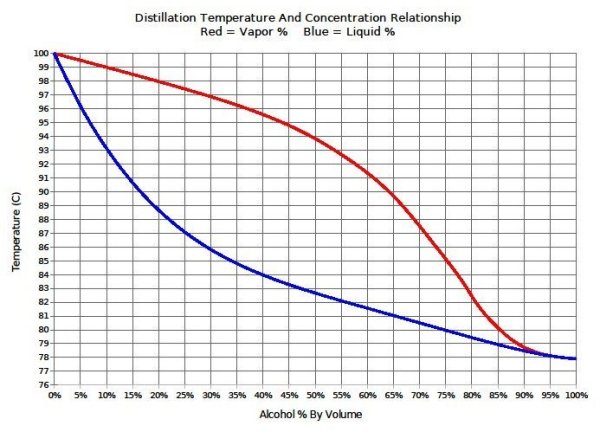
A benzene-methanol system shows azeotropic behavior. After solving for the boiling points for both molecules, describe the behavior for a mixture that is initially rich in benzene (90%) and then for a

Methanol vapor pressure curve. Markers located at atmospheric pressure... | Download Scientific Diagram

Vapor pressure of methanol and ethanol as a function of temperature... | Download Scientific Diagram

A benzene-methanol system shows azeotropic behavior. Include on the graph the boiling points of pure benzene and pure methanol and the boiling point of the azeotrope. | Homework.Study.com

SOLVED: Methanol (CH3OH) has an enthalpy of vaporization of 35.2 kJ/mol and a normal boiling point of 64.7 degrees C. What is the vapor pressure (in torr) of methanol at 30 degrees

![The organic compound which when mixed with ethyl alcohol, [ethanol], makes a spurious is: The organic compound which when mixed with ethyl alcohol, [ethanol], makes a spurious is:](https://dwes9vv9u0550.cloudfront.net/images/4343504/484dd89a-cabf-459f-b5ad-0dd95800e40d.jpg)