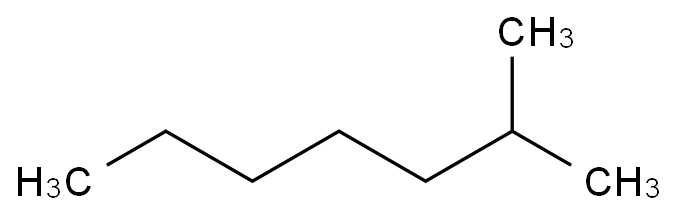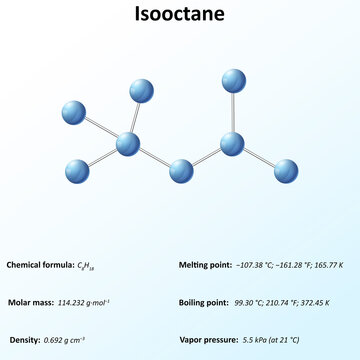
2,2,4-Trimethylpentane, also known as isooctane or iso-octane, is an organic compound with the formula (CH3)3CCH2CH(CH3)2. It is one of several isomers of octane (C8H18) Stock-Illustration | Adobe Stock

p-T diagram of iso-octane with possible fuel conditions for different... | Download Scientific Diagram
![SOLVED: 4. [4 Points]. Gasoline is primarily a comparison mixture of hydrocarbons and sold with an octane rating that is based on a with the combustion properties of isooctane. Gasoline usually contains SOLVED: 4. [4 Points]. Gasoline is primarily a comparison mixture of hydrocarbons and sold with an octane rating that is based on a with the combustion properties of isooctane. Gasoline usually contains](https://cdn.numerade.com/ask_images/c306e15101324e4cb7a3ead962c10943.jpg)
SOLVED: 4. [4 Points]. Gasoline is primarily a comparison mixture of hydrocarbons and sold with an octane rating that is based on a with the combustion properties of isooctane. Gasoline usually contains

Vapor pressure measurements of ethanol–isooctane and 1-butanol–isooctane systems using a new ebulliometer - ScienceDirect

Vapor pressure measurements of ethanol–isooctane and 1-butanol–isooctane systems using a new ebulliometer - ScienceDirect

SOLVED: The normal boiling point of isooctane (C8H18) is 99°C and its enthalpy of vaporization is 35.8 kJ/mol. a) Calculate the vapor pressure of isooctane at 42°C, in mmHg. (You must show
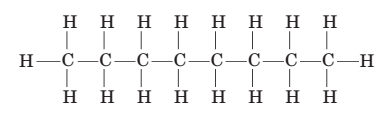
5-118 Isooctane, which has a chemical formula C 8 H 1 8 is the component of gasoline from which the term octane rating derives. (a) Write the balanced chemical equation for the

The role of molecule cluster on the azeotrope and boiling points of isooctane-ethanol blend - ScienceDirect

Volatility characteristics of the fuels used. ULG 78 is a mixture of... | Download Scientific Diagram
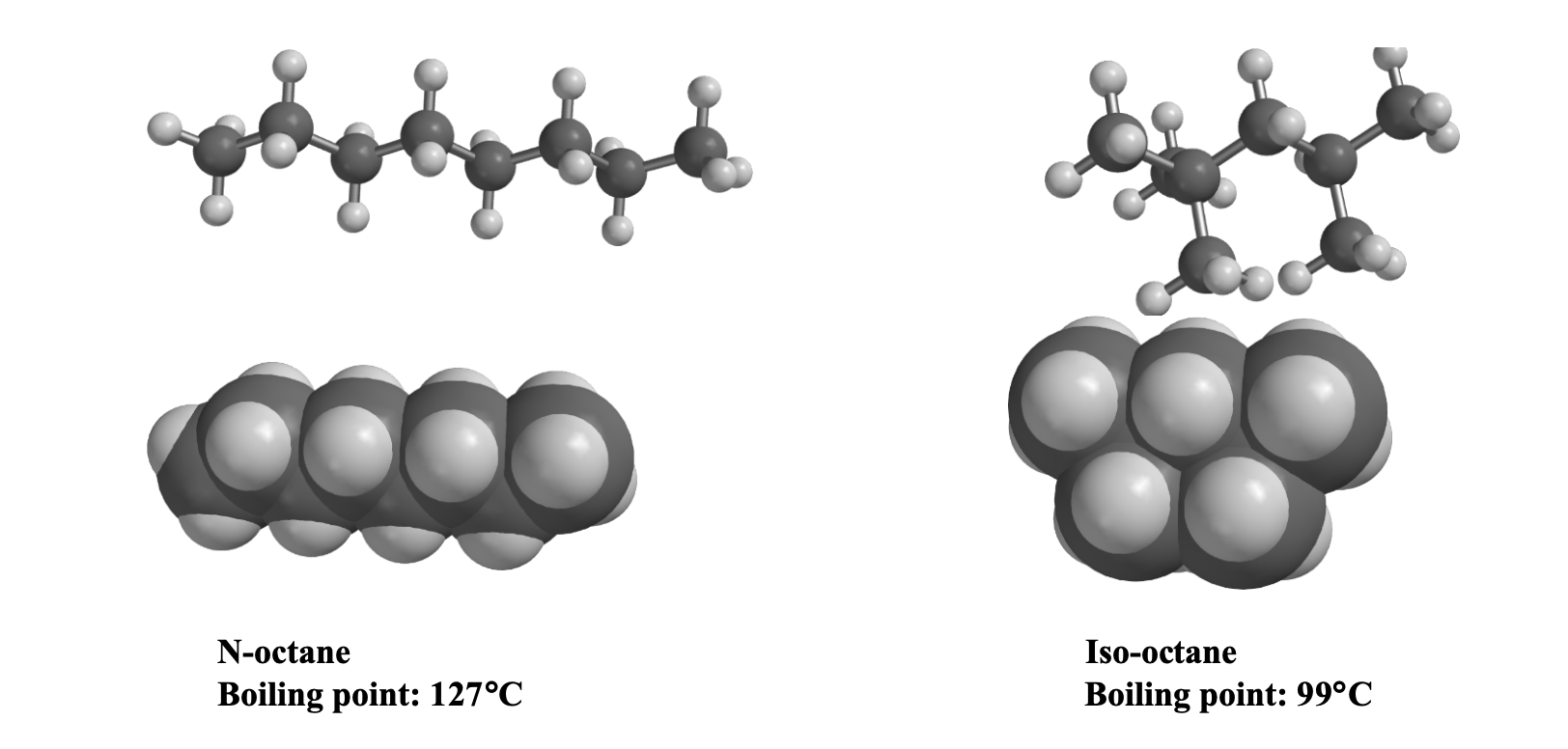
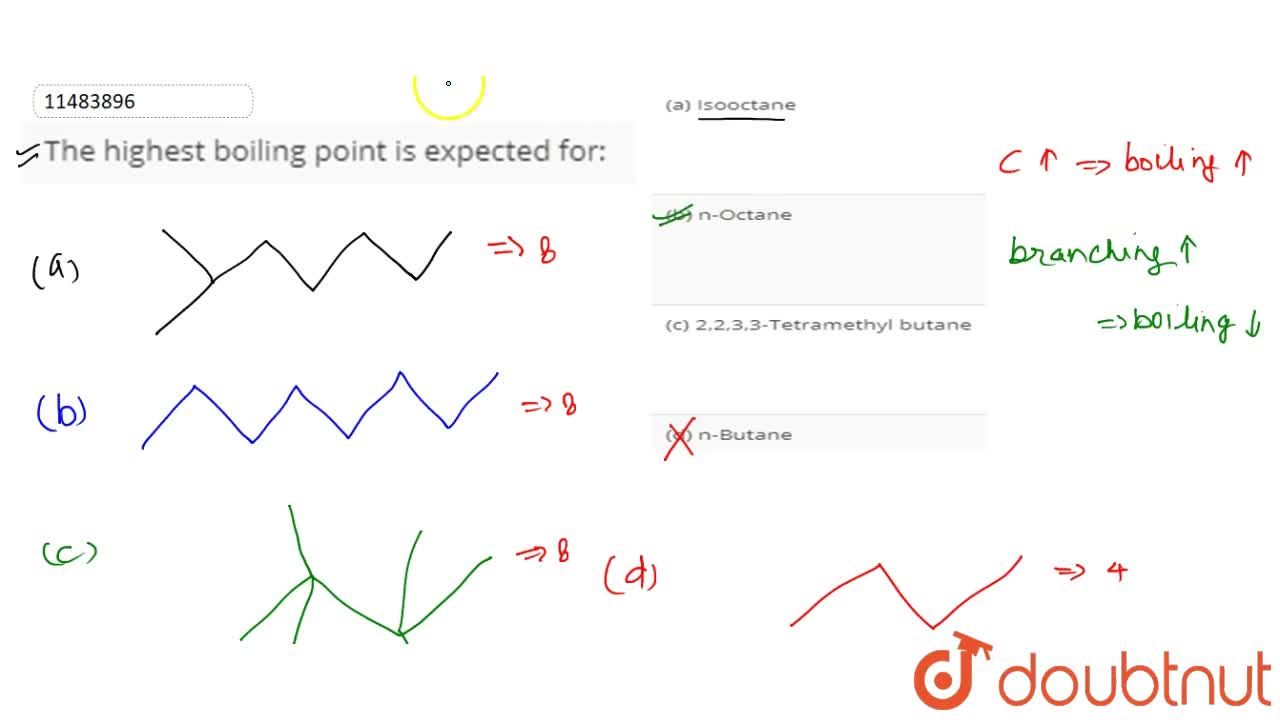
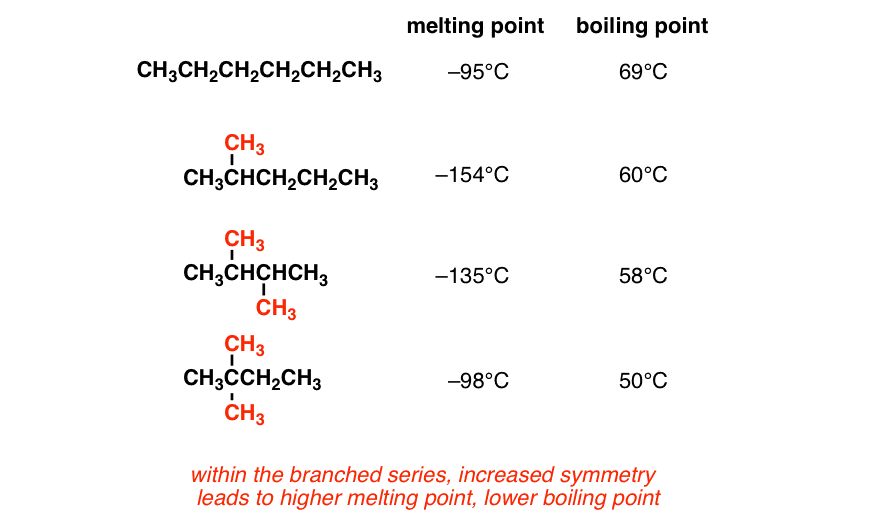
Octane and isooctane are structural isomers with the molecular formula C8H18. The displayed formulas and boiling points of octan

SOLVED: question 36 What factor that best explains the difference in boiling points between octane and isooctane? CH; a CH3CH2CHZCHZCHZCH2CH2CH CH;C-Cza- -CH; CH; octane isooctane b.p. 125.7'C b.p: 99.2*C hydrogen bonding polarizability