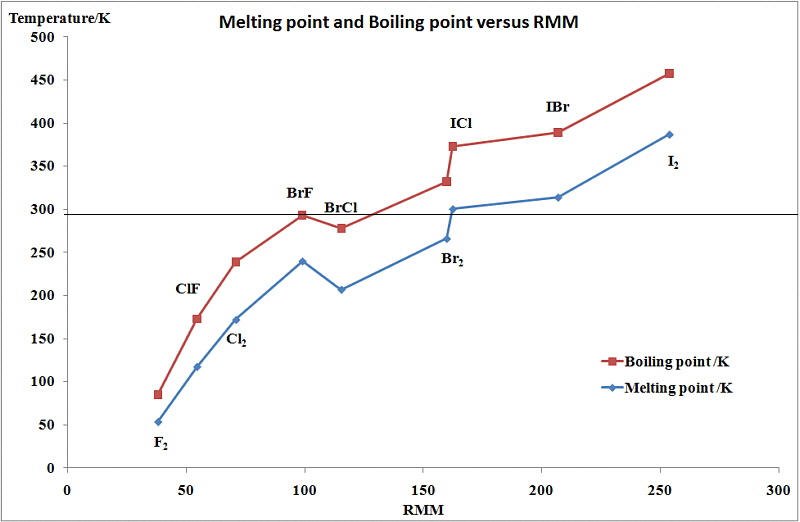
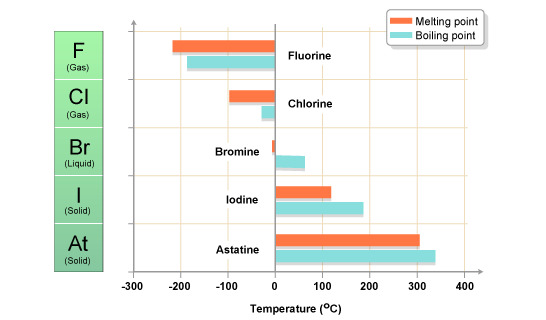
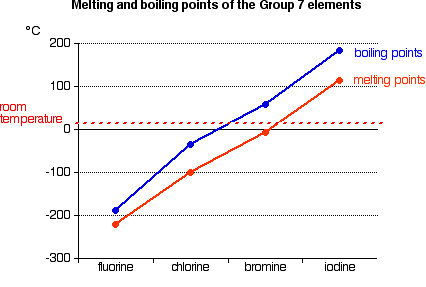
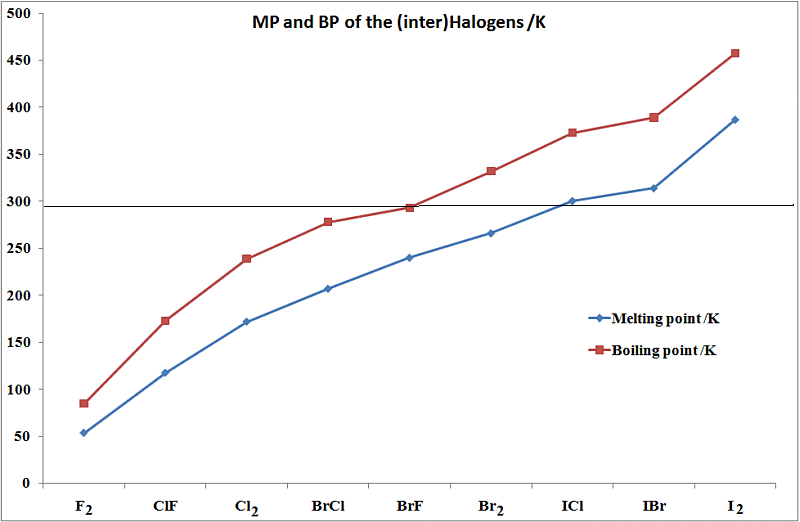
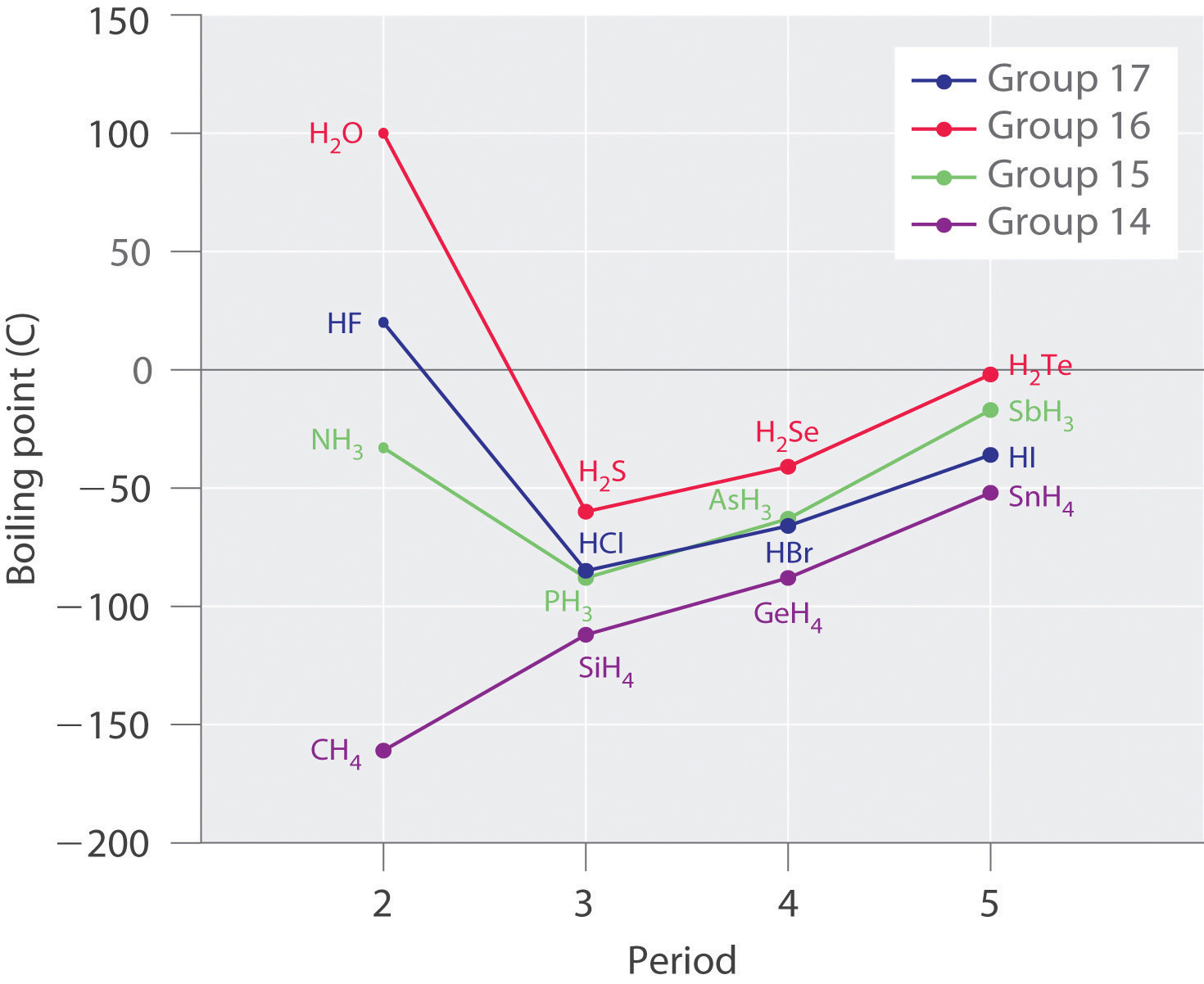
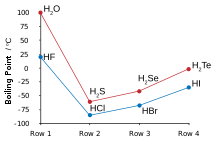
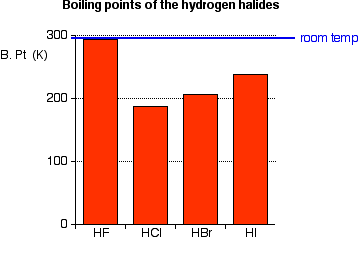
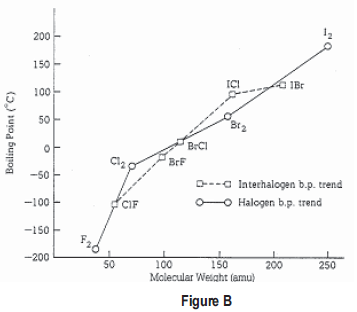
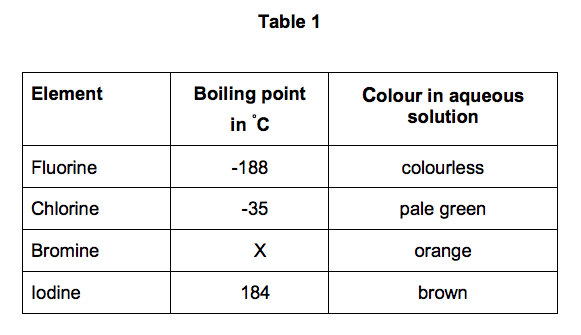
SOLVED: Question 2 The table shows physical properties of halogens. Name Colour Melting point Boiling Pale yellow point 220 188 State at Room Iemperature Gas 2.1.1 101 35 Gas IRed Brown 21.3

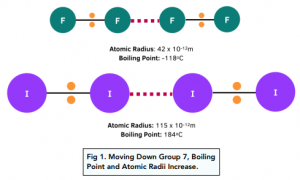
SOLVED: Question 35 (1 point) The boiling points of the halogens increase going from fluorine to iodine. What type of intermolecular force(s) is (are) responsible for this trend? hydrogen bonding ion-ion attraction