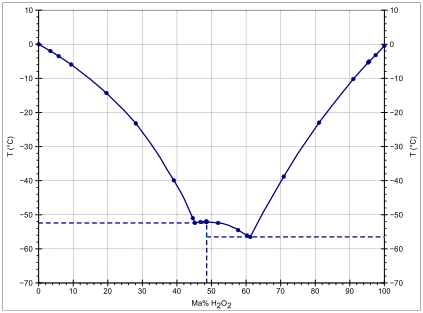
SOLVED: The temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide: H20z(0) = HzOzlg) Use the 298 K thermodynamic information given below
Sciencemadness Discussion Board - Hydrogen Peroxide - Illustrated Practical Guide - Powered by XMB 1.9.11

a) Mention conjugate base of each of the following: HS^-,H3O^+,H2PO4^-,HSO4^-,HF,CH3COOH,C6H5OH,HClO4,NH4^+ (b) Mention the conjugate acid of each of the following: OH^-,CH3COO^-,Cl^-,CO3^2 - ,H2PO4^-,CH3NH2,CH3COOH,NH2^- (c) Which of the following ...

SOLVED:Direct measurement of the normal boiling point of hydrogen peroxide is not possible because pure H2 O2 explodes on heating. The boiling point can be estimated, however, from vapor-pressure data. Use the

Effect of temperature and H2O2 concentration on the carboxyl groups'... | Download Scientific Diagram