Production of a germline-humanized cetuximab scFv and evaluation of its activity in recognizing EGFR- overexpressing cancer cell

Long noncoding RNA and mRNA profiling in cetuximab‐resistant colorectal cancer cells by RNA sequencing analysis - Jing - 2019 - Cancer Medicine - Wiley Online Library

Application of nano-surface and molecular-orientation limited proteolysis to LC–MS bioanalysis of cetuximab | Bioanalysis

Cetuximab–Polymersome–Mertansine Nanodrug for Potent and Targeted Therapy of EGFR-Positive Cancers | Biomacromolecules
Structure–function relationships of the variable domains of monoclonal antibodies approved for cancer treatment

Frontiers | Distinguishing Features of Cetuximab and Panitumumab in Colorectal Cancer and Other Solid Tumors

Schematic representation of trastuzumab and cetuximab. Blue parts of... | Download Scientific Diagram

Different responses of colorectal cancer cells to alternative sequences of cetuximab and oxaliplatin | Scientific Reports
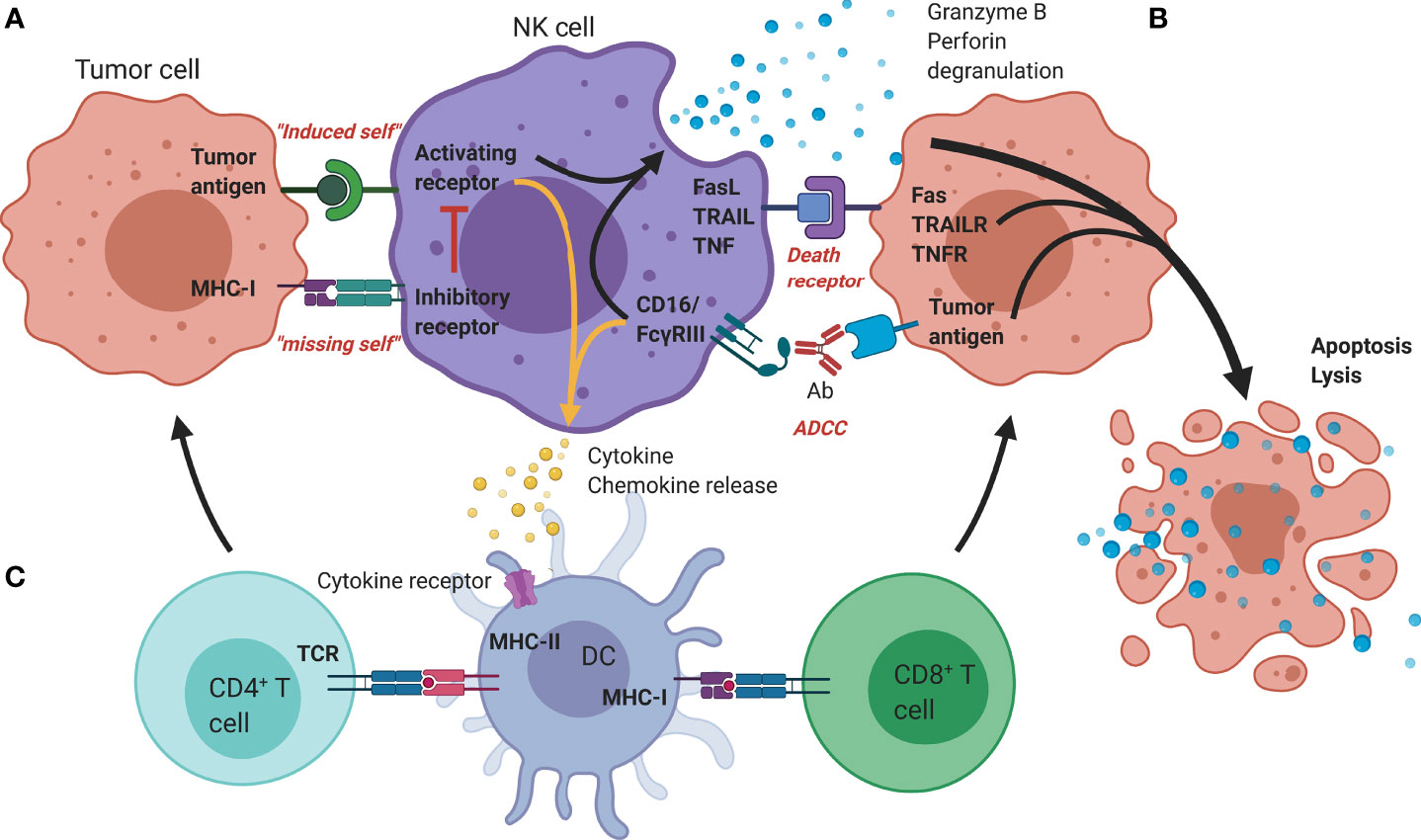
Frontiers | The Right Partner in Crime: Unlocking the Potential of the Anti-EGFR Antibody Cetuximab via Combination With Natural Killer Cell Chartering Immunotherapeutic Strategies

NHRI researchers reports the effect of cetuximab-based sequential therapy as a primary treatment in advanced oral squamous cell carcinoma – NHRI Communications
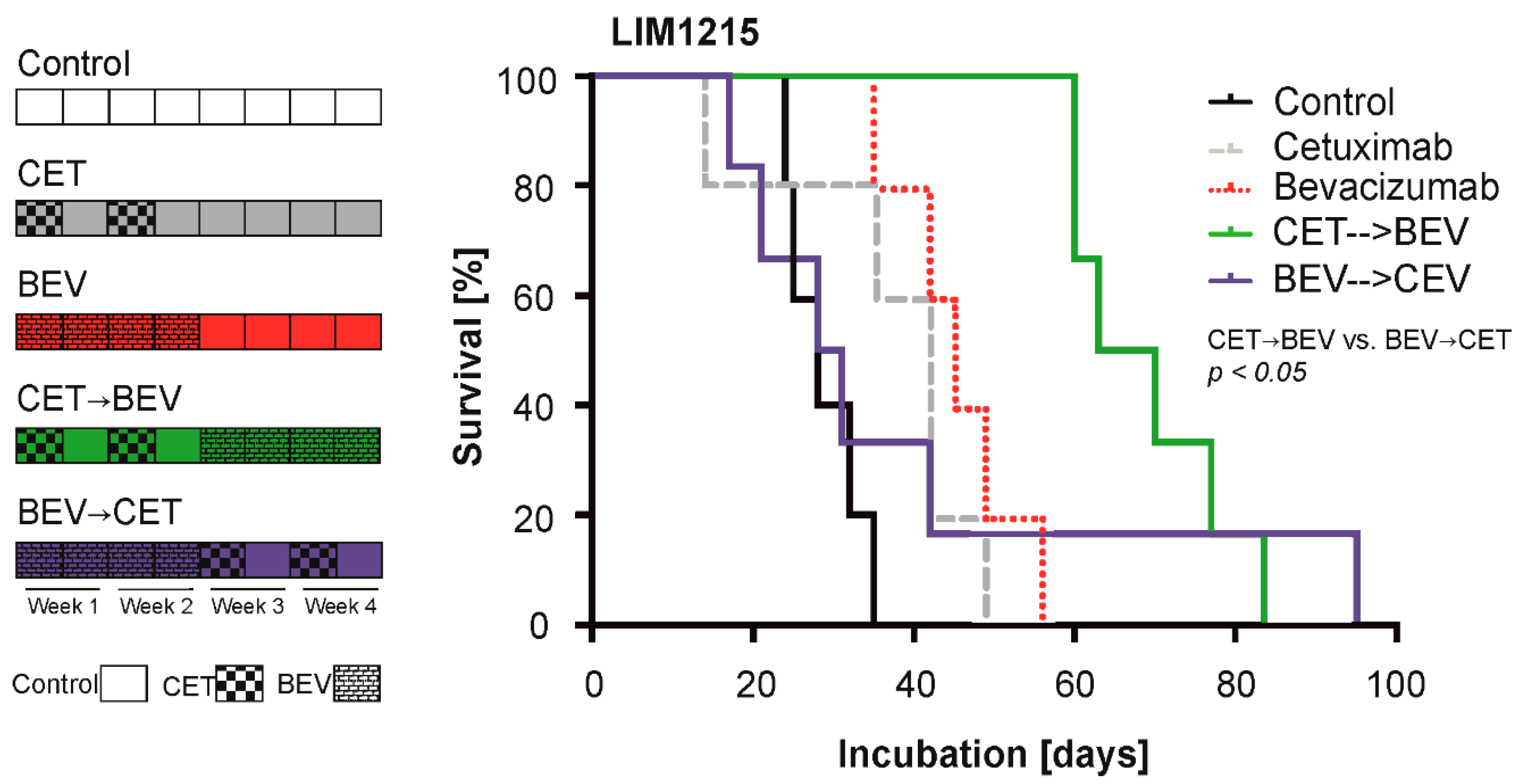
Cancers | Free Full-Text | Cetuximab-Mediated Protection from Hypoxia- Induced Cell Death: Implications for Therapy Sequence in Colorectal Cancer
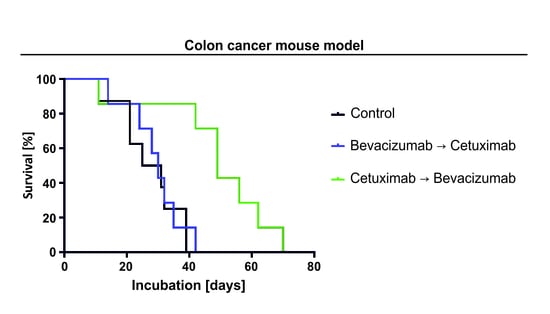
Cancers | Free Full-Text | Cetuximab-Mediated Protection from Hypoxia- Induced Cell Death: Implications for Therapy Sequence in Colorectal Cancer

From combinatorial peptide selection to drug prototype (II): Targeting the epidermal growth factor receptor pathway | PNAS

In silico design, construction and cloning of Trastuzumab humanized monoclonal antibody: A possible biosimilar for Herceptin Akbarzadeh-Sharbaf S, Yakhchali B, Minuchehr Z, Shokrgozar MA, Zeinali S - Adv Biomed Res

Enhancing bacterial production of a recombinant cetuximab-Fab by partial humanization and its utility for drug conjugation - ScienceDirect










