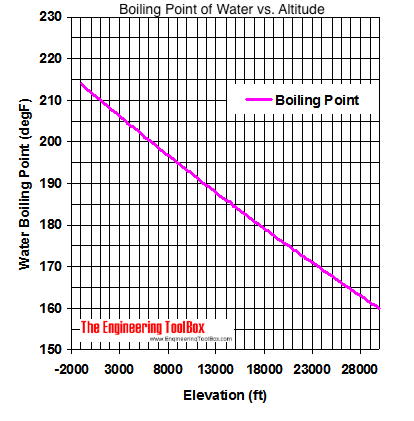
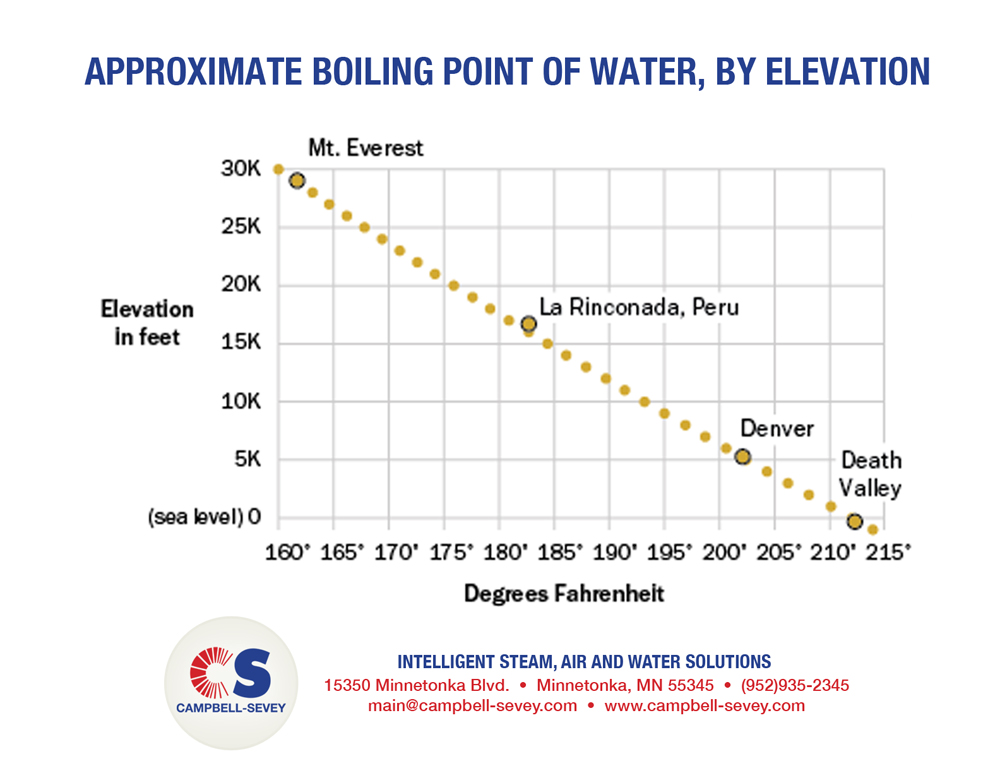
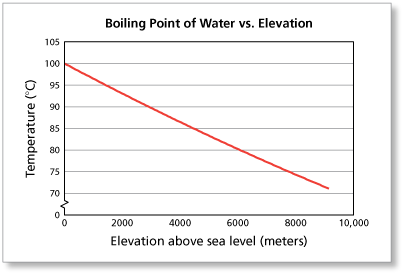
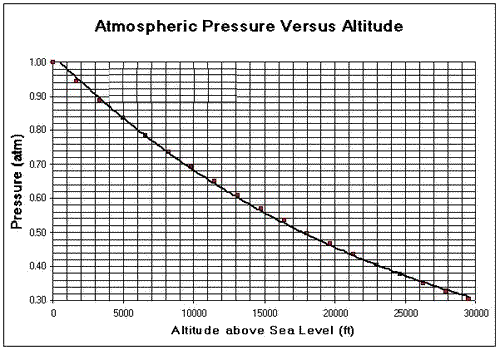
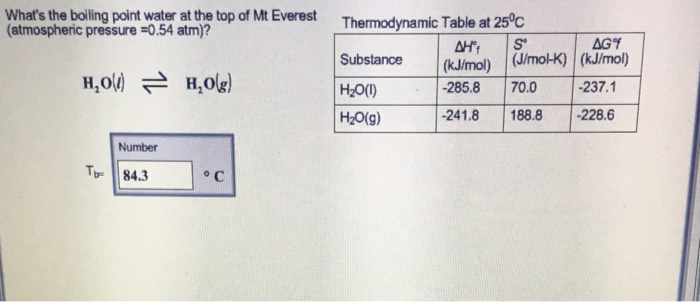
SOLVED: The summit of mount Everest is at 29,029 feet At this elevation the atmosphere pressure is 30 kPa or about 0.30 atm. What is the boiling point of' mount Evcrest? water

What temperature does water boils at? #boilingpoint #water #bp #mounteverest #kilimanjaro #deadsea #sealevel #t… | Water boiling, Sea level, Science and technology

Facts | News | Amazing Stuff on Instagram: “What is the boiling temperature of water on Mount Everest?” | Everest, Mount everest, Water

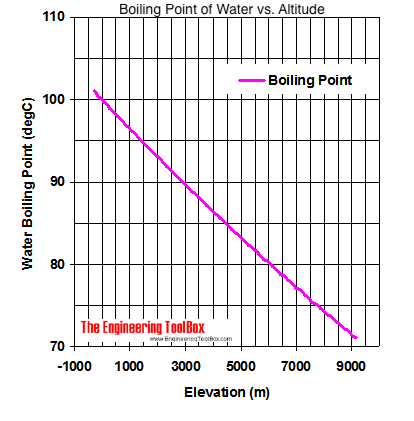
Universal science facts - Why does water boil at 71 degrees on Everest? Liquids start boiling when their vapour pressure equals atmospheric pressur. Atmospheric pressure go on reducing as we go higher

SOLVED:a. Atmospheric pressure on Mount Everest is 224 mm Hg. What is the boiling point of water there? b. What is the freezing point of water on Mount Everest?