BOILING POINT OF A PURE SUBSTANCE Directions: Graph each table of temperature readings below. Plot the time - Brainly.ph

If the normal boiling point of acetone is 56^∘ C and it has a Δ H^∘vap of 32.1 kJ/mol, estimate the boiling point at 5 bar?

SOLVED:The normal boiling point of acetone, an important laboratory and industrial solvent, is 56.2^∘ C and its ΔHvap is 25.5 kJmol^-1 . At what temperature does acetone have a vapor pressure of 375 mmHg ?
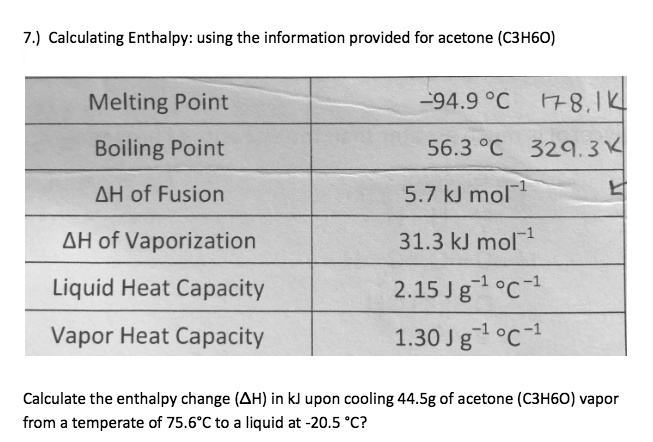
SOLVED: 7.) Calculating Enthalpy: using the information provided for acetone (C3HGO) Melting Point Boiling Point AH of Fusion 94.9 %€ +7-8,1444 56.3 "C 329.34 5.7 kJ mol-1 AH of Vaporization 31.3 kJ

SOLVED: The normal boiling point of acetone, an important laboratory and industrial solvent, is 56.2 °C and its ∆Hvap is 25.5 kJ/mol. At what temperature does acetone have a vapor pressure of

The boiling point of pure acetone is 56.38^(@)C`. When 0.707 g of a compound is dissolved in 10 g of - YouTube
Is a 50/50 mixture of acetone and water an azeotrope? Also, why does the first drop of destillate form at 40C? - Quora

![PDF] Separation of Water-Acetone Mixture Using Suitable Entrainer (Simulation) | Semantic Scholar PDF] Separation of Water-Acetone Mixture Using Suitable Entrainer (Simulation) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3b24cb3fdaba29b0182f3943cf35a512430d1196/29-Table3-2-1.png)