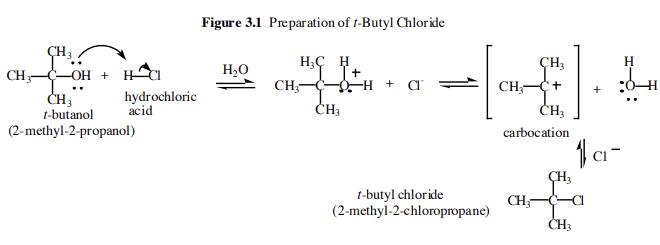
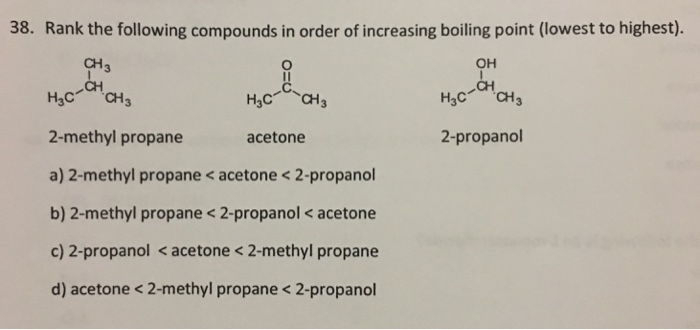
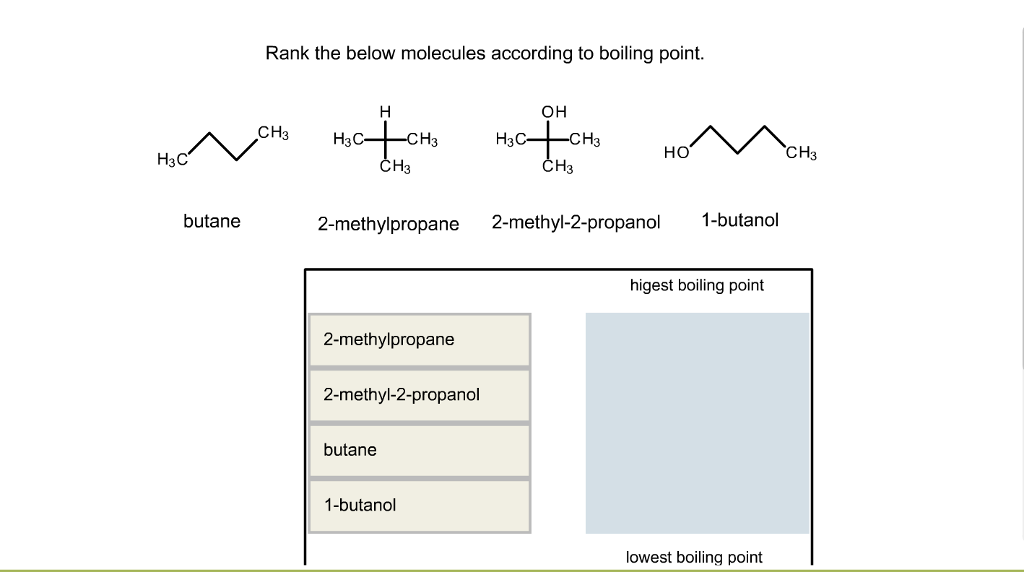
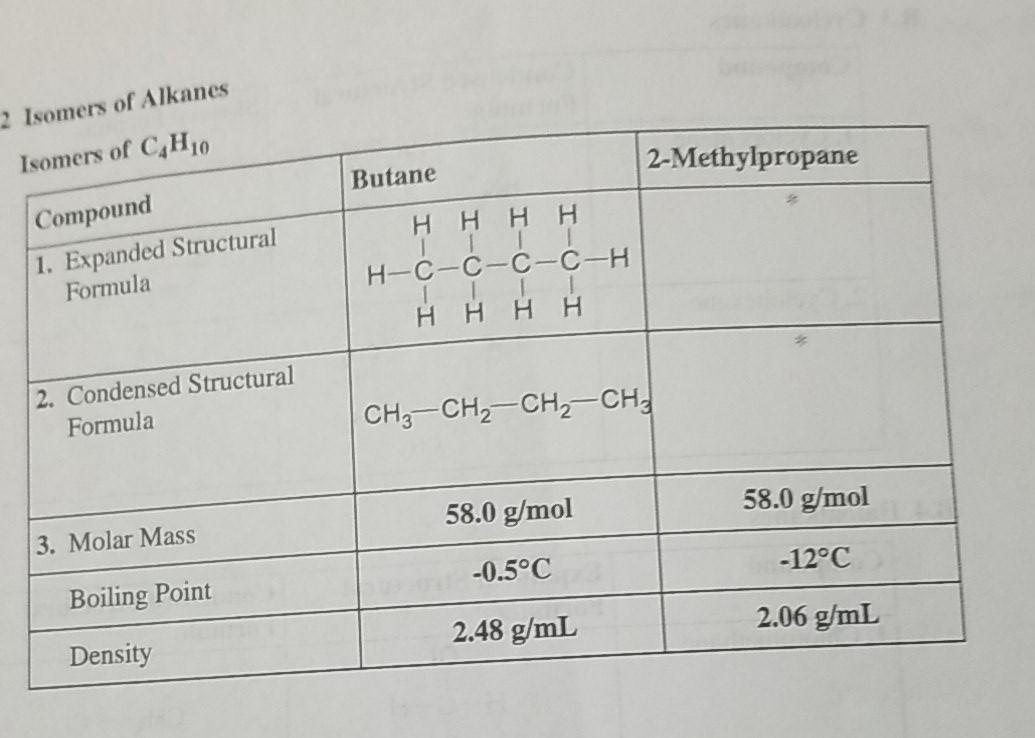
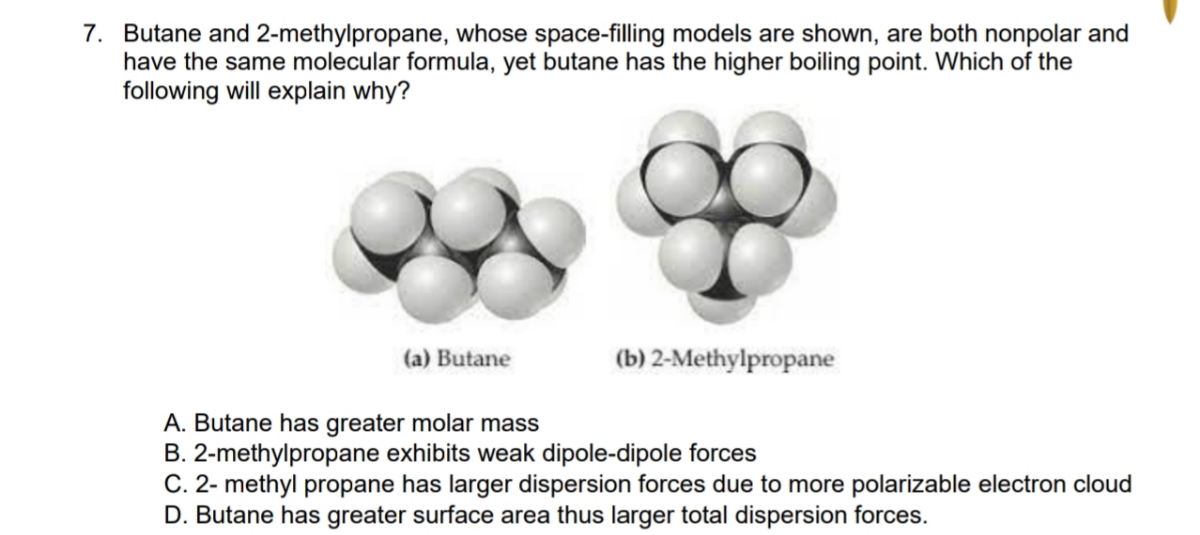
32.Which of the following has the lowest melting point? A)2 methyl butane B)2 methyl propane C)2,2 dimethyl propane D)n pentane

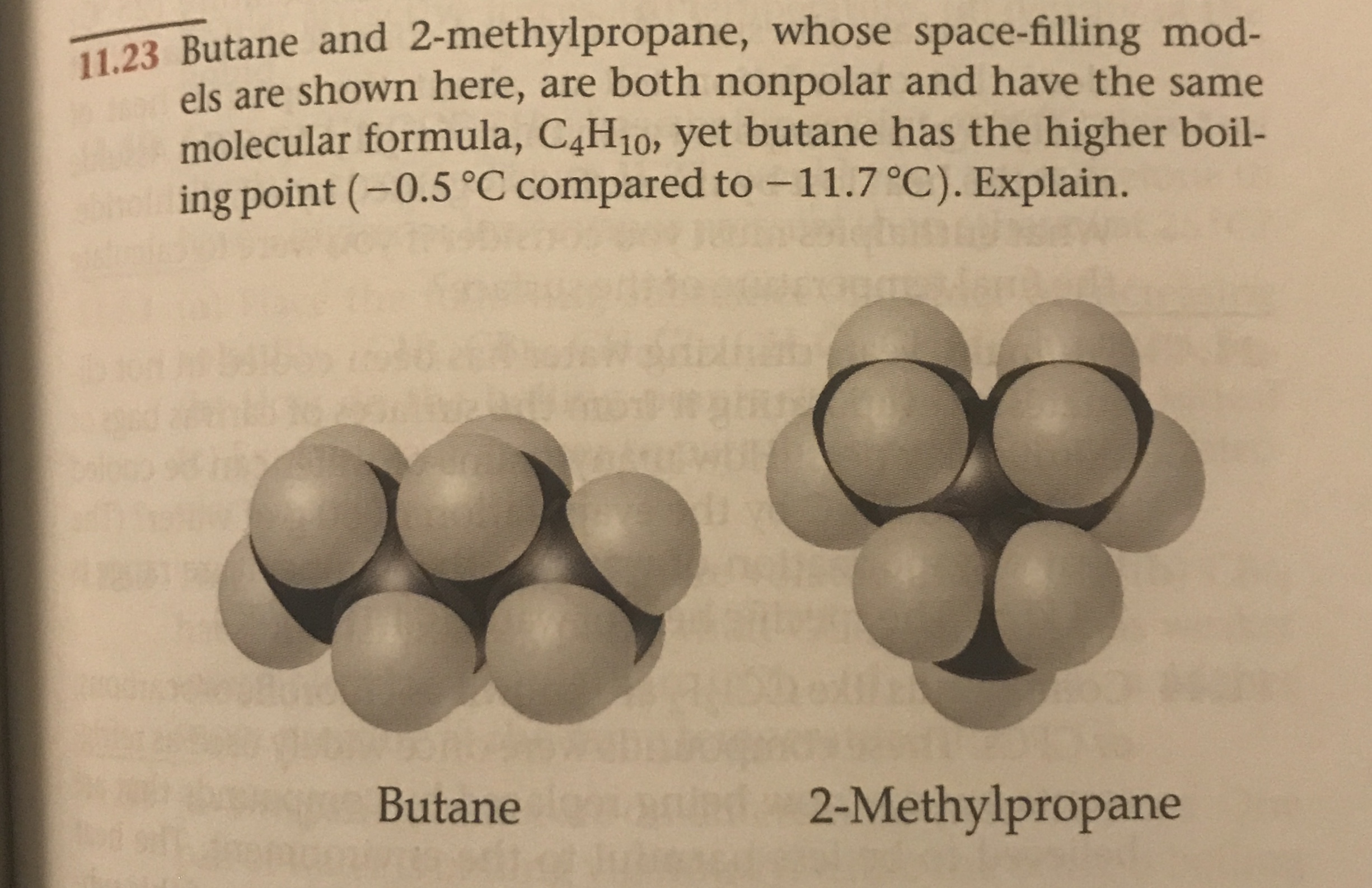
SOLVED: Butane and 2-methylpropane, whose space Hlling models are shown here are both nonpolar and have the same molecular formula C,H,o, yet butane has the higher boiling Point 0.5 C compared t0 -

Unit 13: Organic Chemistry RB Topic 10 I. Organic chemistry - the study of CARBON and carbon compounds - MILLIONS of organic compounds…WHY? the Carbon. - ppt download